 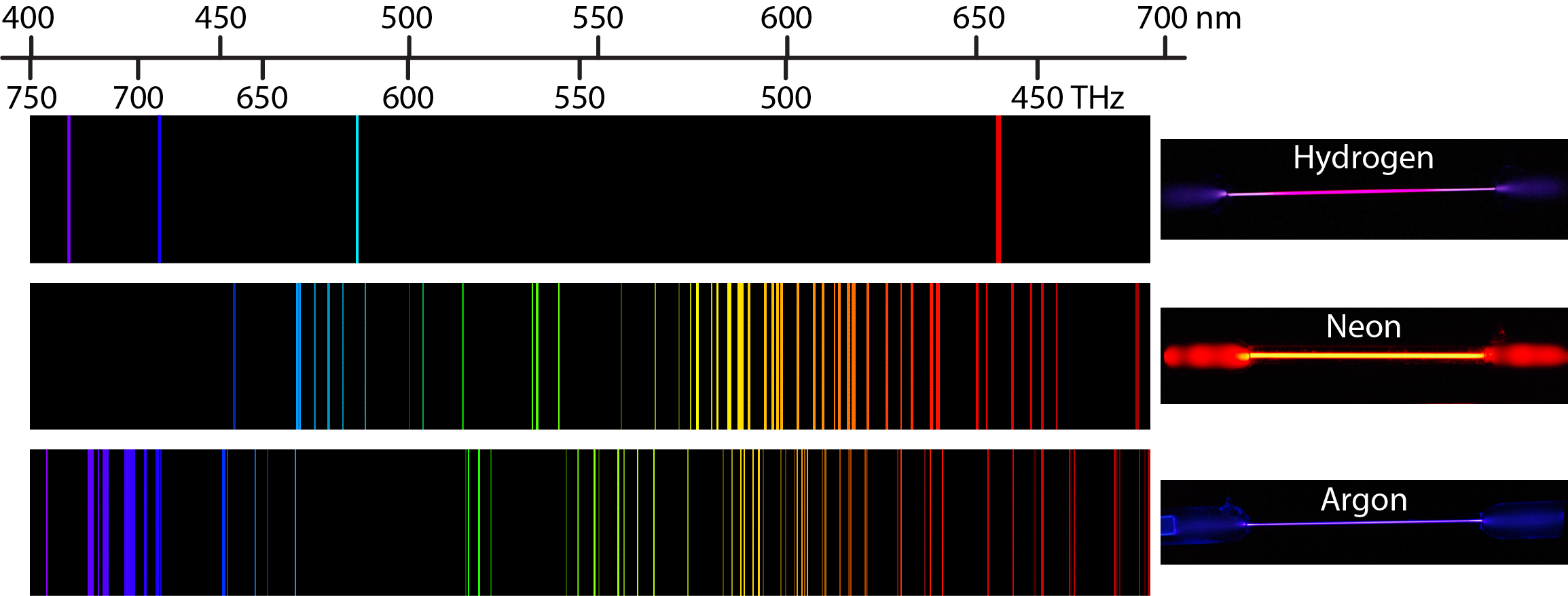
The plasmas used in atomic emission are formed by ionizing a flowing stream of argon, producing argon ions and electrons. Older atomic emission instruments often used a total consumption burner in which the sample is drawn through a capillary tube and in- jected directly into the flame.Ī plasma consists of a hot, partially ionized gas, containing an abundant concentration of cations and electrons that make the plasma a conductor. The burner head consists of single or multiple slots or a Meker-style burner. Flame SourcesĪtomization and excitation in flame atomic emission is accom- plished using the same nebulization and spray chamber assembly used in atomic absorption (see Figure 10.38). Solid samples may be analyzed by dissolving in solution and using a flame or plasma atomizer. The most common methods are flames and plasmas, both of which are useful for liquid or solution samples. The same source of thermal energy usually serves as the excitation source. Atomization and ExcitationĪtomic emission requires a means for converting an analyte in solid, liquid, or solution form to a free gaseous atom. However, are dedicated instruments designed to take advantage of features unique to atomic emission, including the use of plasmas, arcs, sparks, and lasers, as atomization and excitation sources and have an enhancedĬapability for multielemental analysis. Ily adapted for use as flame atomic emission spectrometers by turning off the hol- low cathode lamp and monitoring the difference between the intensity of radiationĮmitted when aspirating the sample and that emitted when aspirating a blank. In fact, most flame atomic absorption spectrometers are eas. I think this is when white light is used that you get an Absorption Spectra.Instrumentation for atomic emission spectroscopy is similar in design to that used for atomic absorption. All the colors of the Absorption Spectra do make it kind of confusing. And these are being absorbed (with emphasis on blue). Actually, if you just burned hydrogen and looked at its spectra, you would get the Emission Spectra and not the Absorption Spectra, and this Emission Spectra would only show the bunch of blue lines, one purple line, and one red line. All the other colors shown are just part of the natural light being shown down on the element. This is the color that will be the opposite of the flame color on the color wheel. Remember, always look at the color area on the rainbow that is blacked out the most. So if blue is being absorbed, the opposite color would be transmitted and this color is orange. However, there are MORE dark lines in the blue region. If you look at the lines for hydrogen blue, purple, and red are being absorbed. Therefore, all the other colors would be absorbed. (This would be orange.) The element hydrogen turns orange when being burned and this color is transmitted to us. 
This means that if there is a big dark band where blue would be, then the opposite color to blue on the color wheel is being transmitted. You are supposed to look at the dark areas of the absorption spectra and those dark areas indicate that the color which would be there is being absorbed. I think both the absorption and emission lines are showing which colors are being absorbed. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
